Follow us on Google News (click on ☆)
A team from Columbia Engineering presents an alternative named S3E. This technique uses a solvent that responds to temperature variations to capture lithium directly from brines, even when its concentration is modest or when it is accompanied by other minerals.

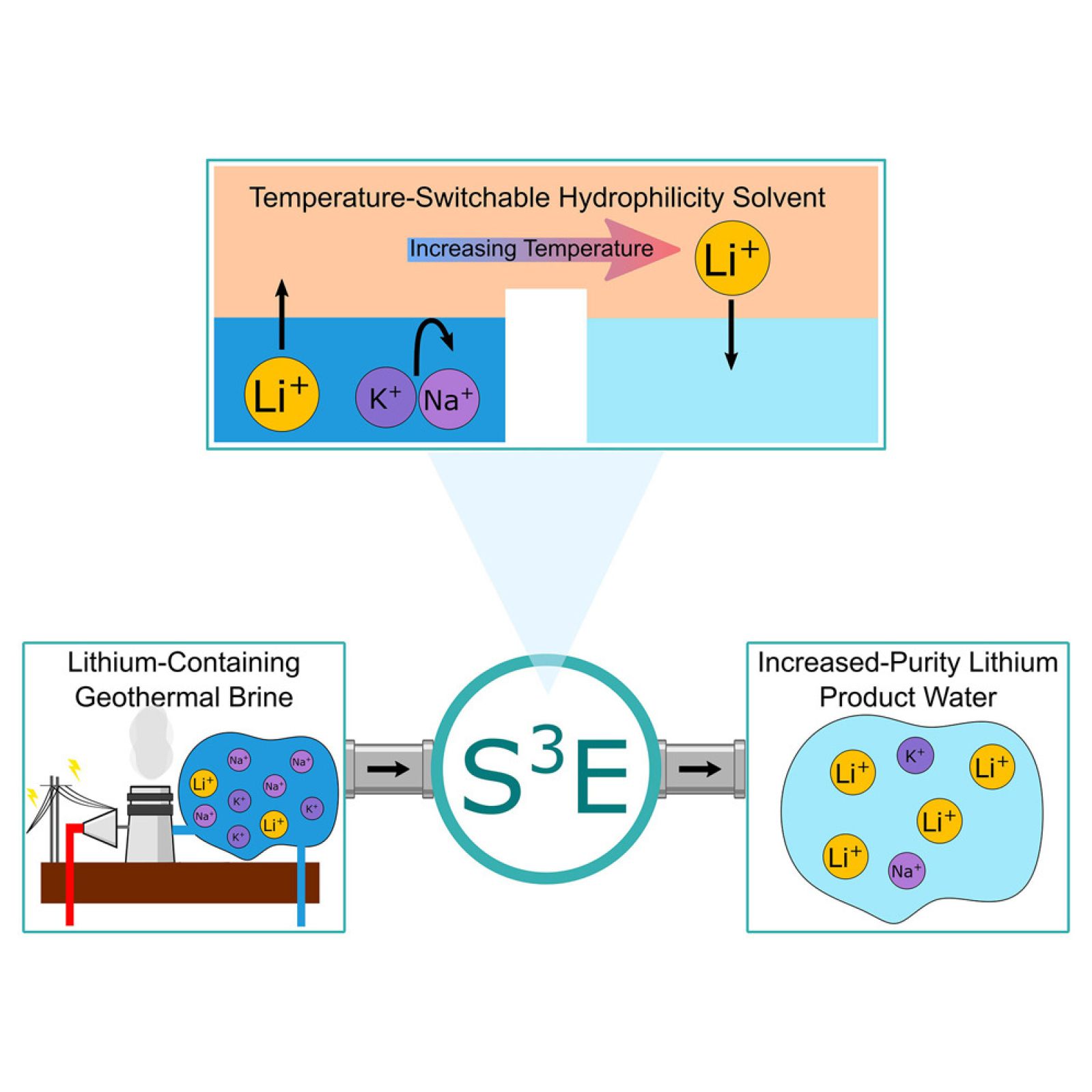
The S3E process works in two main steps. At ambient temperature, the solvent captures the lithium and water present in the brine. Then, during moderate heating, it releases the purified lithium while regenerating itself for reuse. This cyclicity allows for continuous operation to be envisioned.
Unlike solar evaporation, which currently dominates lithium extraction from brines, the S3E method does not depend on specific climatic conditions. It could thus be deployed almost anywhere, such as in regions like the Salton Sea in California, where reserves are abundant but inaccessible with conventional techniques.
Laboratory tests, conducted with synthetic brines modeled on those from the Salton Sea, have shown encouraging results. After only four cycles, nearly 40% of the lithium was recovered using the same batch of solvent. This performance indicates potential for larger-scale operations.
According to the scientists, this approach is selective, fast, and easy to adapt. It can be powered by low-temperature heat from clean energy sources. Ngai Yin Yip, a professor at Columbia University, explained that this technology represents a step towards more sustainable supply chains.
Although this study is still at the proof-of-concept stage, it paves the way for practical applications. By enabling lithium to be extracted more quickly and with less environmental impact, techniques like S3E could contribute significantly to the global energy transition.
The mechanism of the thermally responsive solvent
The solvent used in the S3E technique is designed to change its properties with temperature. When cold, it forms complexes with lithium ions, isolating them from other cations present in the brine. When heated, these complexes dissociate, releasing pure lithium. This cycle can be repeated without loss of efficiency, which lowers operational costs.
The solvent's selectivity is a major advantage. It strongly prefers lithium over sodium or potassium, with ratios up to 10 times higher. Furthermore, it can precipitate magnesium, a frequent contaminant, thus simplifying purification.
The use of low-temperature heat makes the process energy-efficient. This heat can come from renewable or waste recovery sources, aligning the method with sustainability principles. Adjustments could further improve the yield in the future.
This approach relies on fine chemistry that avoids costly additives. By exploiting simple molecular interactions, it offers a robust alternative to traditional processes, with potential applications in other areas of chemical separation.