Follow us on Google News (click on ☆)
How can we destroy cancer cells without damaging healthy tissues? This is one of the main challenges in oncology. Using synthetic DNA strands, a team from the University of Geneva (UNIGE) has created an "intelligent" system capable of recognizing cancer cells with exceptional accuracy and releasing powerful drugs only where they are needed.

Beyond cancer treatment, this research paves the way for a new generation of autonomous and self-regulating drugs. This work is published in Nature Biotechnology.
The ability to directly target tumor cells with specialized drugs is revolutionizing cancer treatment. It preserves healthy tissues and reduces the side effects of chemotherapy. Among the most promising treatments are antibody-drug conjugates (ADCs), which use monoclonal antibodies to deliver drugs directly to cancer cells. Although extremely effective, ADCs suffer from poor tissue penetration and a limited drug-carrying capacity.
To address this, scientists have developed a new technology based on synthetic DNA strands. Due to their small size, DNA components can move through tumors more easily than traditional antibody-based therapies, which are often larger and limited in the amount of drug molecules they can carry.
More targeted and more effective
In this new system, independent DNA strands each carry a distinct component: two strands are associated with different cancer markers, while a third is linked to a cytotoxic drug, i.e., a cell-destroying agent. When the two specific cancer markers simultaneously identify their target and bind to it, the different carrier DNA strands self-assemble to trigger the action of the cytotoxic drug.
This assembly allows for the administration of higher drug concentrations, amplified by several DNA fragments that assemble where needed. Similar to two-factor authentication on a banking website, this process only occurs when both cancer markers are present. If one of the markers is absent, the hybridization chain reaction cannot start and the drug remains inactive.
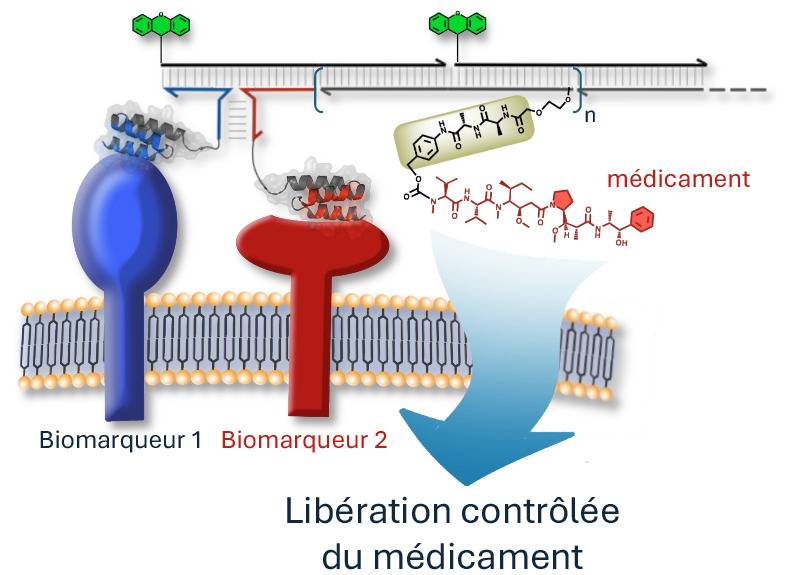
DNA-based smart drugs.
When the two "detectors" simultaneously recognize biomarkers 1 (blue) and 2 (red) on the surface of a diseased cell, the DNA fragments assemble like a puzzle and trigger a chain reaction that releases the drug into the core of that cell, while largely preserving nearby healthy cells.
Adapted from figure 1 of the original article.
In the laboratory, this technology successfully identified cancer cells with specific combinations of surface proteins and selectively administered powerful drugs without damaging neighboring healthy cells. The scientists also demonstrated that it is possible to combine several active molecules within the same treatment, a strategy that could help prevent or overcome drug resistance.
"This could mark a significant advance in the evolution of medicine, with the introduction of autonomous drugs. Until now, computers and AI have helped us design new drugs. The novelty here is that the drug itself can, in a simple way, 'compute' and intelligently respond to biological signals," explains Nicolas Winssinger, full professor in the Department of Organic Chemistry of the Section of Chemistry and Biochemistry at the Faculty of Science of UNIGE, and the last author of this study.
Like a “computer”
Just as computers are built on simple logical operations (AND, OR, NOT), this technology applies the same principle at the molecular level. In this first demonstration, an "AND" logic gate ensures activation only when two cancer biomarkers are present, making the drug highly selective.
Future treatments could integrate more logic functions, allowing them to adapt to the unique and complex response of each patient and minimize side effects. Rather than replacing human supervision, these innovations aim to make therapies more targeted and more effective, thus offering new hope for personalized care.
This research was supported by the Swiss National Science Foundation (SNSF) and builds on the fundamental work of the previous NCCR Chemical Biology program.