Follow us on Google News (click on ☆)
Tardigrades are microscopic animals that live in all moist environments, from the ocean floor to the summit of the Himalayas. Invisible to the naked eye, these organisms also called "water bears" are capable of withstanding extreme stresses such as the vacuum of space, very high pressures, extreme temperatures, or radiation.
Considered the most resilient living beings on Earth, these tiny animals measuring less than 0.04 inches (1 mm) spark the curiosity of scientists who study their remarkable properties and their 41.1% of unique genes that do not exist in any other family of living organisms.
Thus, the protein Dsup (for Damage suppressor), named after one of these unique genes from which it originates, is capable of protecting tardigrades' DNA from oxidative stress, and particularly from radiation and free radicals. This discovery had already been successfully exploited to make human cells more resistant to radiation.
But a question arose from this study: what would happen if Dsup was expressed in a whole organism? To answer this, chemists and biologists from the CNRS, INSERM, and the University of Montpellier introduced the Dsup gene into the genome of Caenorhabditis elegans (C. elegans), a tiny worm measuring 0.04 inches (1 mm) widely used as a quantitative genetics model, among other things in research on aging.
Using a precise gene insertion method, they generated worms expressing Dsup without altering their growth, fertility, or behavior. These worms proved resistant to X-rays and oxidative stressors like hydrogen peroxide. Dsup seems to act not by activating the classic cellular defense mechanisms, but by directly reducing the amount of free radicals, those unstable molecules that damage cells.
The most astonishing thing: the worms expressing Dsup live longer. And this, without triggering the usual genetic pathways linked to longevity, like that of the FOXO/DAF-16 protein, nor disrupting reproduction. Even better, the Montpellier team observed that Dsup decreases oxygen consumption by mitochondria, the cell's power plants, without altering their structure, suggesting a fine modulation of metabolism.
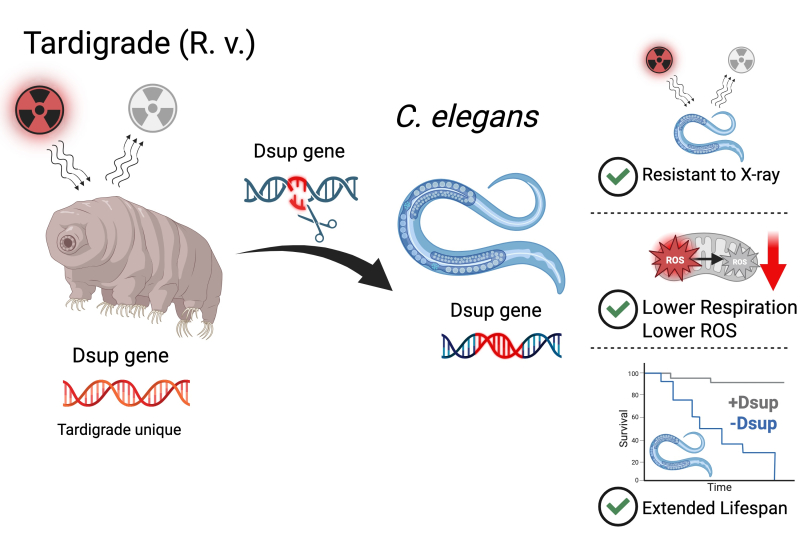
A specimen of the tiny worm C. elegans can be genetically modified by introducing the Dsup gene from the tardigrade into its genome. The Dsup protein expressed by this gene in the worm protects its DNA, reduces oxidative stress, and extends its lifespan. A unique tardigrade gene, from elsewhere, with powerful effects on cellular survival.
© Simon Galas, Myriam Richaud & Aymeric Bailly
This work, published in the journal Science Advances, opens a new path in the study of aging. For the first time, introducing a gene from another species allows for the extension of an animal's lifespan without major side effects. Dsup could well be a new tool for understanding, and perhaps one day controlling, the mechanisms of cellular aging.
Editor: AVR
