Follow us on Google News (click on ☆)
To manufacture its own molecules without being detected, the virus steals a protective element belonging to the cell's RNA. Work described in the journal Nature finally details this molecular hijacking.

Influenza viruses possess a genome made of RNA, a molecule similar to DNA that notably serves as a blueprint for producing proteins. When a virus enters a cell, it cannot reproduce on its own. It therefore hijacks the cell's machinery to manufacture its own copies.
But a problem arises for the virus. Cells know how to recognize suspicious RNA and then trigger an antiviral response. To avoid this alert, influenza viruses use a trick: they snatch a small protective "tag" present on some cellular RNA.
This tag, called a "cap," is found at the beginning of messenger RNAs produced by the cell. It acts somewhat like an identification badge. Thanks to it, the RNA is stable, can be used to make proteins, and is recognized as belonging to the cell.
Without this cap, RNA is quickly considered foreign. The cell can then activate defense mechanisms to block the infection. Many viruses have their own tools to make this cap. Influenza viruses, however, have chosen another solution: stealing it.
Their main enzyme, called viral polymerase or FluPol, comes close to the cellular machinery that makes RNA, RNA polymerase II. Researchers knew these two machines interacted, but the precise sequence of the hijacking remained unclear.
To understand this mechanism, several scientific teams recreated the phenomenon in the lab. They then observed the proteins involved using cryo-electron microscopy, a technique capable of visualizing molecular structures with very high precision.
The observations show that the virus acts in three steps. First, its polymerase attaches near the cellular machinery just as a new RNA is being manufactured and has just received its cap.
Then, the viral enzyme cuts this RNA very close to this cap. The virus then recovers a very short fragment of RNA that already has the protective tag. This small piece is then used as a starting point to manufacture a viral RNA. Result: the RNA produced by the virus also possesses a cap and closely resembles a normal cellular RNA. The cell therefore has more difficulty detecting it.
The researchers also identified the importance of a cellular factor called DSIF, which participates in forming the complex to which the viral polymerase attaches. Experiments show that if the contact between the viral polymerase and cellular proteins is disrupted, viral RNA production decreases sharply.
This suggests an interesting avenue for future drugs. Blocking this interaction could prevent the virus from stealing the cap from cellular RNAs and thus slow the infection.
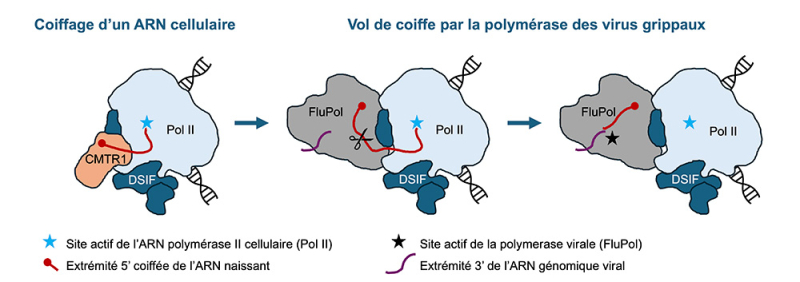
Model illustrating the "cap-snatching" performed by influenza virus polymerases (FluPol).
During the manufacture of an RNA by the cellular polymerase (Pol II), a protective cap is added to its end. The viral polymerase then attaches to the cellular complex and cuts the RNA near this cap. The virus recovers this short capped fragment and uses it as a starting point to manufacture a viral RNA that also possesses a cap.
© Nadia Naffakh