Follow us on Google News (click on ☆)
But this robustness has a downside: once molded and cured, they can no longer be remelted or recycled.
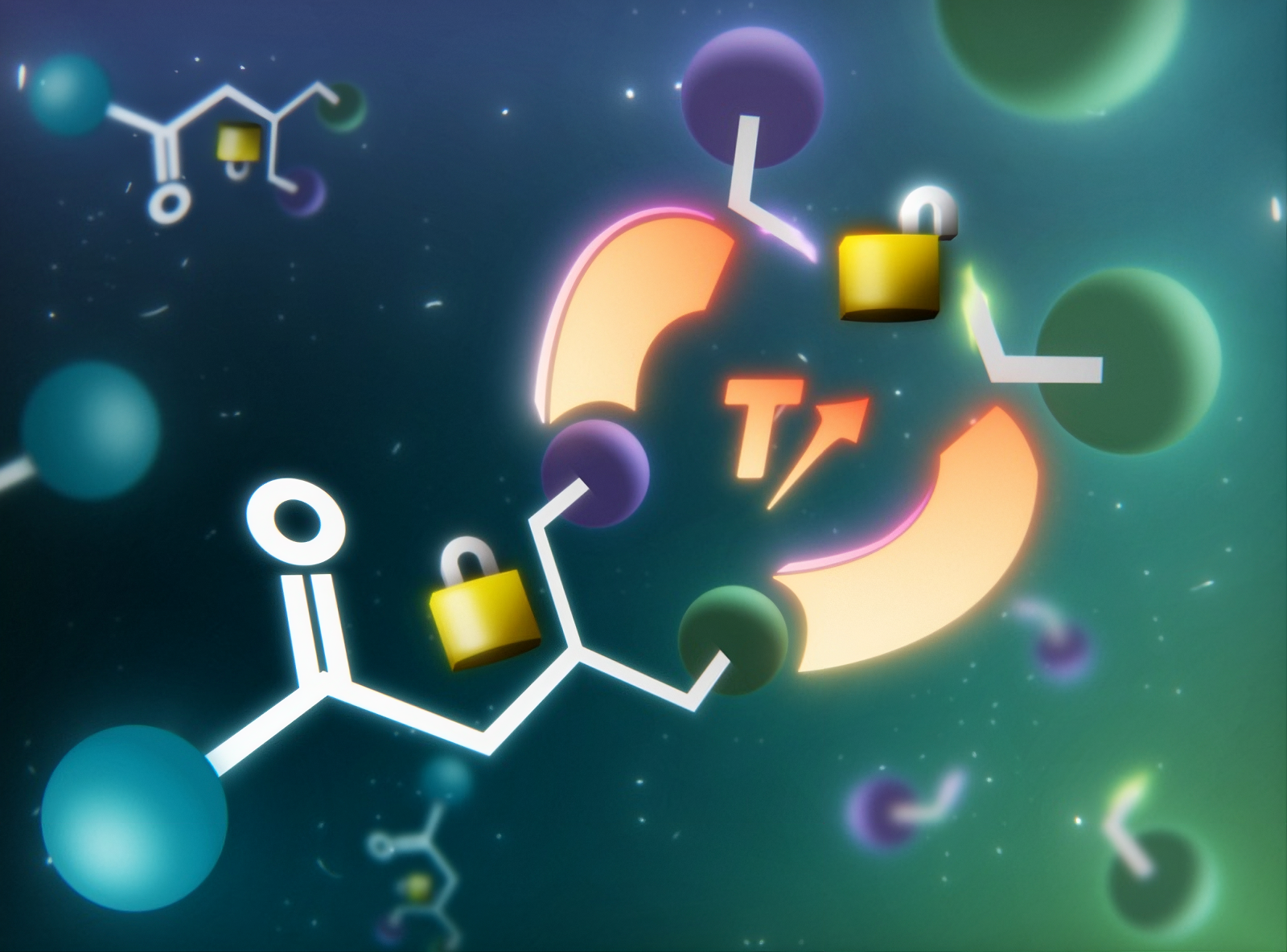
For about a decade, adaptable covalent networks (CANs) have been generating significant interest. Indeed, these next-generation plastics combine the robustness of thermoset materials with the possibility of being reshaped and/or recycled. This property relies on so-called "dynamic" chemical bonds, capable of breaking and reforming under a stimulus, such as temperature.
Until now, these rearrangements required the presence of highly reactive chemical groups in excess, particularly sulfur-containing groups called thiols. But in return, this excess makes the material more sensitive to "creep," a phenomenon of slow and irreversible deformation under prolonged stress that ultimately compromises the material's durability and mechanical performance.
To overcome this difficulty, scientists at the Softmat Laboratory (CNRS/University of Toulouse) have developed an original strategy akin to a molecular "lock." Their idea: temporarily mask the excess thiol groups responsible for creep to prevent their reactivity at operating temperature, while preserving the possibility of reactivating them when needed.
They thus succeeded in protecting the sulfur atoms of the thiol groups within stable chemical structures that act as locked locks. At room temperature, these locks prevent any unwanted interaction between the sulfur atoms and their environment. The material remains stable, resistant, and less prone to creep.
Once heated, these locks gradually open, releasing the thiol groups which can then participate in exchange reactions allowing the material to be reshaped or recycled. This strategy thus makes it possible to thermally activate or deactivate the material's recyclability while limiting creep at operating temperature.
This advancement, which is the subject of a publication in the journal Polymer Chemistry, opens new perspectives for the development of more durable plastics with an extended lifespan while remaining recyclable. Beyond that, this strategy could be extended to the design of materials capable of adapting their properties according to their conditions of use.