Follow us on Google News (click on ☆)
Reactions that seem to defy the laws of thermodynamics
Life possesses a remarkable property: it manages to maintain and exploit chemical states far from equilibrium. As early as 1945, physicist Erwin Schrödinger pondered this particularity in his book What is Life?, noting that living organisms take advantage of energy gradients to organize matter.
A central example is chemiosmosis, a mechanism used by cells to produce energy. In mitochondria or bacteria, a proton gradient across a membrane enables the generation of chemical reactions essential for metabolism.
But a question remains: could similar processes exist in purely mineral environments, even before the appearance of the first cells?
Mineral "membranes" capable of producing metallic iron
To explore this hypothesis, scientists reproduced laboratory analogues of iron-rich alkaline hydrothermal vents, similar to those that existed on early Earth.
The results, published in the journal Nature Communications, reveal an unexpected phenomenon: the energy of a pH gradient can be converted into the production of metallic iron, a reaction that, in principle, should not occur without the presence of a chemical reducing agent.
This process relies on coupled transfers of ions and electrons within mineral "membranes." These structures then act as true energy interfaces, comparable in some aspects to biological membranes.
Locally, these transfers allow for the maintenance of non-equilibrium redox states, associated with a local decrease in entropy, a condition considered essential for the emergence of primitive metabolic reactions.
An unexpected mechanism
The results challenge some previous hypotheses. Until now, it was thought that this type of reaction mainly depended on reactive chemical compounds generated by pH differences.
However, the study shows that the intrinsic properties of the minerals are sufficient: the very structure of the mineral membrane allows the direct conversion of the energy from the pH gradient into a redox gradient, essential to the chemical reactions found in living systems.
This discovery suggests that purely geological systems could have played an active role in the first prebiotic energy conversions.
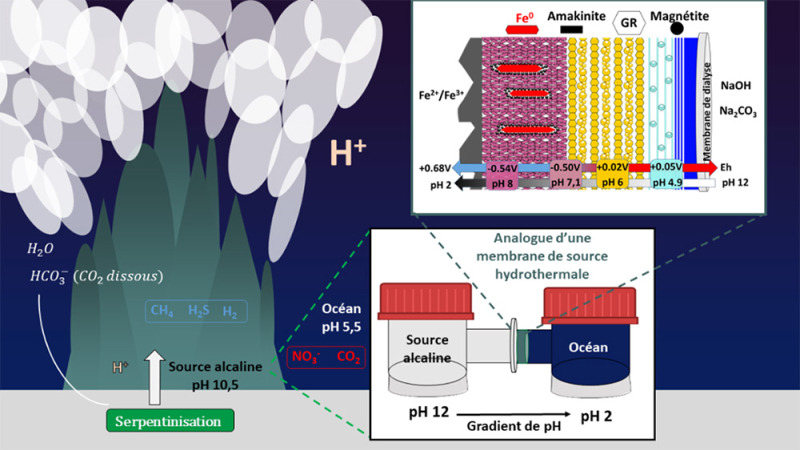
Experimental reconstruction of an analogue of the mineral membrane from alkaline hydrothermal vents (AHV).
The precipitation of iron oxyhydroxides at the interface between an alkaline solution and an acidic solution leads to the formation of a structured mineral barrier.
This membrane generates pH and redox potential gradients, associated with the dismutation of Fe²⁺ into Fe³⁺ (magnetite) and Fe⁰, which evokes a mechanism analogous to living mechanisms, locally producing redox states out of thermodynamic equilibrium.
© Simon Duval
Toward a reconstruction of the first steps of metabolism
This study suggests that geological environments, like hydrothermal vents, could have played a key role in catalyzing prebiotic reactions. It also provides an experimental model for studying bioenergetic processes that still function in modern cells. It could also explain previously unexplained geochemical transformations in natural environments.
In the future, scientists plan to explore more complex reactions in this type of setup, notably the condensation of phosphate units, to get closer to cellular processes.
This is another step toward reconstructing the conditions that allowed the emergence of the common ancestor of all life forms on Earth.
