Follow us on Google News (click on ☆)
Two teams from Inserm, CNRS and Université Paris Cité have just discovered the conductor of this mechanism of inexorable progression: the HNF1B protein. Beyond a major advance in the understanding of these diseases, the teams have identified a new therapeutic target of major interest. This work is to be published in the journal Science.
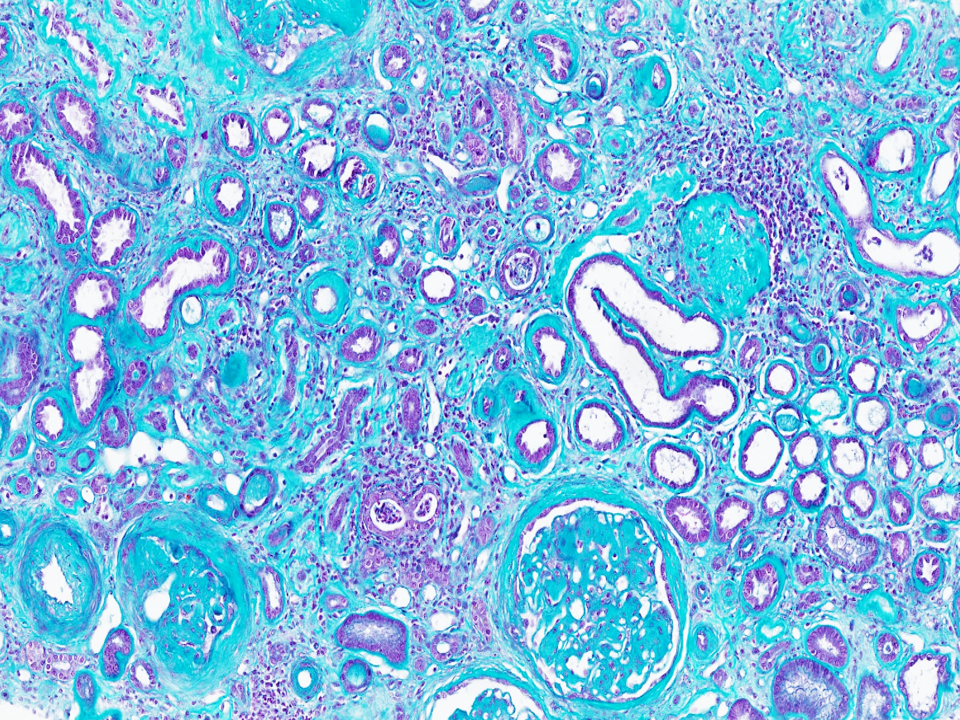
Light microscopy image of a kidney from a patient with end-stage renal failure. Significant interstitial fibrosis (collagen and fibrotic connective tissue in cyan) and atrophy of the renal tubules (small ducts surrounded by a thickened wall in purple) indicate a gradual and irreversible replacement of functional kidney tissue with scar tissue.
Staining: Masson's trichrome.
© Fabiola Terzi, Marco Pontoglio/Inserm
Chronic kidney disease affects more than 10% of the world's population, or about 850 million people. It is characterized by a progressive and irreversible loss of kidney function, which may require dialysis or even transplantation.
Several risk factors are well identified: diabetes, hypertension, obesity or inflammation. However, even if the initial cause is treated, the decline in kidney function is, in the vast majority of cases, unavoidable. This self-perpetuating nature represents a particularly worrying aspect of kidney disease; by acquiring its own dynamic, it becomes difficult to stop. Until now, the biological mechanisms responsible for this progression remained largely unknown.
Two teams from the Institut Necker-Enfants malades (Inserm/CNRS/Université Paris Cité) led by Marco Pontoglio, CNRS research director, and Fabiola Terzi, Inserm research director and director of the Institut Necker-Enfants malades, have studied these biological mechanisms, and more specifically the role of the HNF1B protein (Hepatocyte Nuclear Factor 1 beta) in the adult kidney. It controls the expression of many genes and plays an essential role in kidney formation during embryonic development.
Indeed, in humans, mutations in the HNF1B gene cause a decrease in the activity of this protein, which leads to a rare genetic kidney disease. And strikingly: the lesions observed in patients can resemble those found in the most common forms of chronic kidney disease, namely fibrosis or atrophy of kidney tissue.
This similarity led the researchers to formulate a hypothesis: the dysfunction of HNF1B could represent a common mechanism linking rare kidney diseases and the much more frequent forms such as chronic kidney disease.
The research teams thus observed that the loss of HNF1B activity in the adult human and mouse kidney caused rapid and severe chronic kidney failure, accompanied by fibrosis and atrophy of kidney tissue. Deprived of HNF1B activity, the renal tubular cells - which line the renal tubules and play a major role in kidney function - normally highly differentiated and stable, lost their identity and specialization and began to proliferate inappropriately.
This proliferation was then accompanied by cell death or premature cellular aging, contributing to progressive kidney fibrosis and deterioration of its function.
The researchers also identified a set of genes whose expression is regulated by HNF1B. This set appeared altered very early in several mouse models of chronic kidney disease, sometimes even before the appearance of visible lesions, and was associated with a defect in kidney tissue repair.
The scientists also showed that factors associated with kidney disease, such as inflammation or the presence of albumin in the urine, reduced HNF1B activity. Using advanced techniques for analyzing gene activity, they highlighted a key phenomenon: the stress undergone by kidney cells during chronic kidney disease leads to repression of genes regulated by HNF1B.
"Our results reveal the existence of a true vicious cycle that explains the self-perpetuating nature of kidney diseases: the decrease in HNF1B activity would promote kidney disease, and in return, kidney disease would progressively suppress HNF1B activity, further worsening kidney damage. This mechanism could explain why chronic kidney disease tends to worsen continuously, even in the absence of new aggression," summarizes Marco Pontoglio.
This mechanism would be common to many kidney diseases. Indeed, analysis of more than 900 kidney biopsies from patients with chronic kidney disease of different origins and covering all stages of severity systematically highlighted the characteristic molecular signature of loss of HNF1B function.
The abnormalities were all the more marked as the disease was advanced, confirming the major role of this protein in the severity of kidney damage. "This study establishes HNF1B as a true guardian of kidney function. Its loss of activity links for the first time rare genetic kidney diseases and common forms of chronic kidney disease through a single mechanism," clarifies Fabiola Terzi.
These results open a major new therapeutic perspective: "finding a way to restore HNF1B activity could slow down, or even modify the course of chronic kidney disease," concludes Fabiola Terzi.
This work is the subject of a patent filed with Inserm Transfert in 2025.